CRISPR gene editing is no longer a concept confined to the pages of science fiction novels. Today, it stands as the most versatile and precise tool in a scientist’s kit, offering the power to rewrite the very code of life. From curing hereditary diseases to creating sustainable food sources, the impact of CRISPR gene editing is being felt across every sector of biology.
What is CRISPR Gene Editing?
At its simplest, CRISPR gene editing is a technology that functions like a high-tech “search and replace” tool for DNA. CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats. It is a natural defense mechanism found in bacteria, which scientists have repurposed to edit human, animal, and plant genomes with incredible accuracy.
Unlike previous methods of genetic modification that were expensive and time-consuming, CRISPR gene editing is fast, affordable, and highly specific. It allows researchers to target a specific sequence of DNA and make precise cuts, either to disable a harmful gene or to insert a beneficial one.
How CRISPR Gene Editing Works
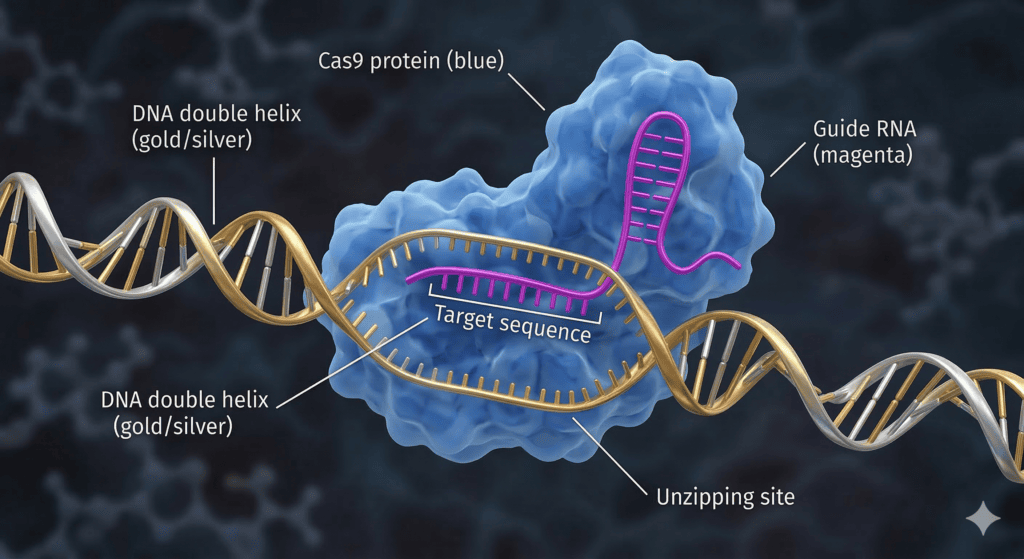
To understand how CRISPR gene editing functions, imagine a pair of molecular scissors guided by a GPS system. The process involves two key components:
- Cas9 Enzyme: This acts as the “scissors” that can cut through the DNA strands.
- Guide RNA (gRNA): This acts as the “GPS” that leads the Cas9 enzyme to the exact location in the genome that needs to be edited.
Once the Cas9 enzyme reaches the target, it makes a clean cut. The cell then recognizes the damage and begins to repair itself. During this repair process, scientists can “trick” the cell into using a new piece of DNA, effectively changing the genetic instructions of the organism. This precision is why CRISPR gene editing is considered a revolutionary leap over older technologies.
7 Real-World Uses of CRISPR Gene Editing
The versatility of this technology means it is being applied in diverse and surprising ways. Here are some of the most impactful real-world examples:
1. Curing Genetic Disorders: The Victory Over Sickle Cell
The most significant milestone for CRISPR gene editing occurred in late 2023 and early 2024 when regulatory bodies (like the FDA) approved the first-ever CRISPR-based therapy, Casgevy.
Sickle cell disease is caused by a single “typo” in the DNA that makes hemoglobin. By using CRISPR gene editing, scientists can go into a patient’s bone marrow cells and “turn on” a different type of hemoglobin—fetal hemoglobin—which we usually stop producing after birth. This fetal hemoglobin doesn’t sickle, effectively bypassing the genetic defect.
In clinical trials, patients who had suffered from “pain crises” their entire lives were suddenly symptom-free. This isn’t just a treatment; for many, it is a functional cure. This success has opened the door for CRISPR gene editing to be tested on other conditions like Beta-thalassemia and Hereditary Transthyretin Amyloidosis.
2. Cancer Immunotherapy: Building “Super Cells”
Standard chemotherapy is often described as a “sledgehammer” approach—it kills cancer, but it hurts the rest of the body too. CRISPR gene editing offers a “scalpel.”
Through a process called CAR-T cell therapy, doctors extract a patient’s own T-cells (the soldiers of the immune system). Using CRISPR gene editing, they delete the genes that allow cancer cells to “hide” from the immune system. They also add new genetic instructions that help the T-cells recognize specific markers on a tumor.
Once these “super cells” are infused back into the patient, they hunt down the cancer with robotic precision. Recent studies in 2025 have shown that CRISPR gene editing can make these cells more durable, allowing them to fight solid tumors—like those found in lung or breast cancer—which were previously very difficult to treat with immunotherapy.
3. Pest Control: Rewriting Entire Ecosystems
One of the most controversial yet impactful uses of CRISPR gene editing is the “Gene Drive.” Normally, a trait has a 50% chance of being passed to offspring. With a CRISPR-powered gene drive, that chance is pushed to nearly 100%.
In regions of Africa and South Asia, malaria remains a leading cause of death. Scientists are using CRISPR gene editing to edit Anopheles mosquitoes so they can no longer carry the malaria parasite. By releasing a few thousand of these edited mosquitoes into the wild, the trait spreads through the entire population within a few generations.
While the potential to save millions of lives is clear, the use of CRISPR gene editing in the wild raises ecological questions. Scientists are currently working on “reversal drives”—a sort of “undo” button for CRISPR gene editing—to ensure we can control these changes if they affect the food chain in unexpected ways.
4. Resilient Agriculture: Feeding 10 Billion People
As the global population climbs toward 10 billion, our current farming methods are under stress. CRISPR gene editing is providing a solution that is faster and more precise than traditional GMOs (Genetically Modified Organisms).
For example, researchers have used CRISPR gene editing to develop:
- Non-browning mushrooms: By “turning off” the gene responsible for oxidation, these mushrooms stay fresh longer, reducing food waste.
- Gluten-reduced wheat: Scientists are editing the complex gluten proteins in wheat to make it safer for people with mild sensitivities.
- Climate-ready rice: By editing genes responsible for salt tolerance, farmers can now grow rice in coastal areas where rising sea levels have made the soil too salty for traditional crops.
Because CRISPR gene editing often just “deletes” a gene rather than adding foreign DNA from another species, many countries are regulating these crops more leniently than old-school GMOs.
5. De-Extinction: Bringing Back the Woolly Mammoth
The “Colossal Biosciences” project has gained international attention for its attempt to use CRISPR gene editing to resurrect the Woolly Mammoth. This isn’t just a vanity project; it’s about environmental restoration.
Mammoths once packed down the snow and knocked over trees in the Arctic, keeping the permafrost cold. Without them, the permafrost is melting and releasing massive amounts of methane. By using CRISPR gene editing to insert mammoth traits (like thick hair and subcutaneous fat) into the Asian Elephant genome, scientists hope to create a “proxy” species that can live in the Arctic and restore the “Mammoth Steppe” ecosystem.
6. Sustainable Energy: Algae as the New Oil
Biofuels have always struggled because growing corn or soy for fuel takes up valuable land needed for food. Algae, however, can grow in plastic tubes in the desert.
Using CRISPR gene editing, synthetic biologists have identified the genes that regulate fat production in algae. By “knocking out” the genes that limit fat storage, they have created strains of algae that are essentially tiny oil factories. This CRISPR gene editing breakthrough could lead to jet fuel that is carbon-neutral, as the algae absorb $CO_2$ as they grow.
7. Xenotransplantation: Solving the Organ Shortage
Every day, people die waiting for a kidney or heart transplant. Pigs have organs that are roughly the same size as humans, but our immune systems immediately reject them. Furthermore, pig DNA contains ancient viruses called PERVs (Porcine Endogenous Retroviruses) that could infect humans.
In a landmark study, researchers used CRISPR gene editing to “clean” the pig genome. They deactivated dozens of viral genes and edited several “human-like” markers into the pig’s DNA. These CRISPR gene editing advancements have already led to the first successful (though experimental) transplants of pig hearts and kidneys into human patients, signaling a future where the organ waiting list no longer exists.
The Ethical Landscape: Where Do We Draw the Line?
As we master CRISPR gene editing, we face the “He Jiankui” dilemma—named after the scientist who illegally edited the genes of twin babies in 2018. While “somatic” editing (editing a patient’s own cells) is widely accepted, “germline” editing (editing embryos so the change is passed to future generations) remains a red line for most of the global scientific community.
The power of CRISPR gene editing requires a global consensus on ethics. We must ensure that this technology doesn’t create a “genetic divide” where only the wealthy can afford to enhance their children’s intelligence or physical traits.
The Business of Biology: The Billion-Dollar Patent War
For over a decade, a fierce legal battle raged between the Broad Institute (MIT/Harvard) and the University of California, Berkeley. While Jennifer Doudna and Emmanuelle Charpentier won the Nobel Prize for discovering the mechanism, the Broad Institute held the patents for using CRISPR gene editing in human cells (eukaryotes).
This conflict created a complex “patent thicket.” Companies like Editas Medicine, CRISPR Therapeutics, and Intellia Therapeutics had to choose sides, licensing technology from different institutions. In 2022 and 2023, major rulings largely favored the Broad Institute, but the ripple effects continue to influence how much CRISPR gene editing therapies cost today. When a single treatment for Sickle Cell costs over $2 million, the debate over who “owns” the code of life becomes more than just academic—it’s a matter of global health equity.
Beyond Cas9: The Rise of Cas12 and Cas13
While Cas9 is the most famous tool in the CRISPR gene editing arsenal, it is not the only one. Scientists have recently unlocked new enzymes that allow for even more specialized tasks.
Cas12: The Precise Cleaner
Cas12 is often preferred over Cas9 for certain types of CRISPR gene editing because it leaves “sticky ends” when it cuts DNA. These staggered cuts make it much easier for scientists to insert new genetic material into the gap. In 2026, Cas12 is being used extensively in agricultural innovation to stack multiple beneficial traits—like drought resistance and high vitamin content—into a single seed.
Cas13: Targeting RNA
While Cas9 and Cas12 edit DNA (the permanent blueprint), Cas13 targets RNA (the temporary messenger). This is a game-changer for medical safety. By using CRISPR gene editing on RNA, scientists can temporarily turn off a disease-causing protein without making a permanent change to the patient’s genetic code. This “reversible” gene editing is currently being tested to treat chronic pain and temporary viral infections, providing a safety net that traditional DNA editing cannot offer.
Real-World Example: Rapid Viral Diagnostics
During the minor respiratory outbreaks of late 2025, Cas13 was the hero of the diagnostic world. Scientists developed a “CRISPR-in-a-box” test that could detect viral RNA in under 20 minutes with the accuracy of a full-scale PCR lab. This application of CRISPR gene editing is revolutionizing how we handle global health security, moving testing from distant hospitals directly into the palms of our hands.
The “Dark Side” and Biosecurity
As CRISPR gene editing becomes more accessible, “biohacking” has moved from high-end labs to garage workshops. This democratization of science is exciting but carries risks.
- Dual-Use Concerns: The same technology used to cure a virus could theoretically be used to make a virus more lethal.
- Off-Target Mutations: Even with Cas9 precision, there is a risk of “genomic instability.” If CRISPR gene editing cuts the wrong spot, it could accidentally trigger a cell to become cancerous.
- The Genetic Divide: There is a fear that CRISPR gene editing will create a new form of inequality—”Gattaca”-style—where only the elite can afford genetic enhancements for strength, memory, or longevity.
Conclusion
The era of CRISPR gene editing is here. It is a tool of unprecedented power, capable of healing the sick, feeding the hungry, and protecting our planet. As we continue to refine the Cas9 protein and explore new variants like “Prime Editing,” the accuracy of CRISPR gene editing will only improve.
We are no longer just observers of evolution; we are its architects. The choices we make with CRISPR gene editing today will determine the biological heritage of the human race for centuries to come.