Herpes simplex virus as an anticancer agent is no longer just a lab idea—it is already being used in real patients and approved for certain types of melanoma. Researchers are now engineering this common virus to selectively kill cancer cells and trigger powerful immune responses against tumors.
In this in‑depth guide, you’ll learn how herpes simplex virus as an anticancer agent works, what the clinical evidence shows, and where this exciting form of oncolytic virotherapy is heading next.
What is oncolytic virotherapy?
Oncolytic virotherapy is a type of cancer treatment that uses live, replication‑competent viruses to selectively infect and destroy cancer cells while sparing most normal tissue. These oncolytic viruses are either naturally tumor‑selective or genetically engineered to prefer tumor cells over healthy cells.
Oncolytic viruses work in two main ways:
- They directly lyse (burst) cancer cells after viral replication inside the tumor.
- They stimulate the immune system by turning the tumor into an in‑situ cancer vaccine, exposing tumor antigens to immune cells.
Herpes simplex virus as an anticancer agent belongs to this broader class of oncolytic viruses, alongside adenovirus, vaccinia, reovirus, and others.
Why herpes simplex virus as an anticancer agent is so promising
Researchers favor herpes simplex virus type 1 (HSV‑1) as an oncolytic backbone for several biological and practical reasons.
Key advantages of HSV‑1 as an oncolytic virus
- Large, engineerable genome
HSV‑1 has about 152 kb of double‑stranded DNA, with roughly 30 kb that can be deleted or replaced by therapeutic genes without losing replication ability. This allows scientists to “arm” herpes simplex virus as an anticancer agent with immune‑stimulating payloads. - Non‑integrating DNA virus
HSV‑1 replicates its DNA in the nucleus but does not integrate into the host genome, which reduces the risk of insertional mutagenesis compared with retroviruses. - Sensitive to antiviral drugs
Standard anti‑herpes drugs like acyclovir and ganciclovir can rapidly shut down HSV replication. This gives clinicians a safety “off switch” when using herpes simplex virus as an anticancer agent. - Broad cell tropism
HSV can infect many cell types, including various epithelial and neural cells, which is helpful when designing therapies for diverse solid tumors. - Well‑understood biology and clinical history
Decades of research on HSV infections (like cold sores) mean that its life cycle, immune interactions, and safety profile are well characterized.
Thanks to these features, herpes simplex virus as an anticancer agent can be precisely modified to be safer, more selective, and more immunogenic than its wild‑type counterpart.
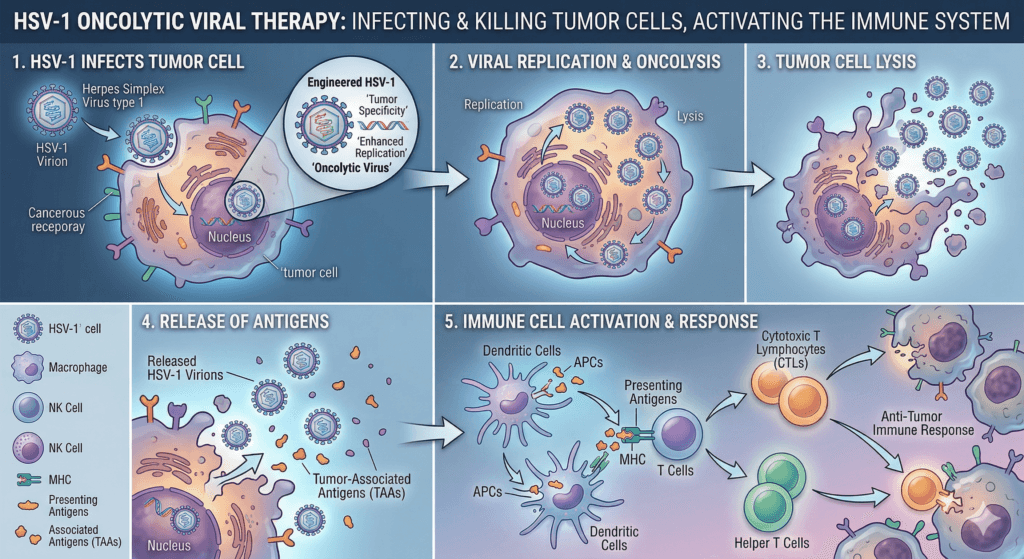
How oncolytic herpes simplex virus kills cancer cells
Herpes simplex virus as an anticancer agent relies on a combination of genetic engineering and tumor‑specific weaknesses to attack cancer.
1. Tumor‑selective replication and lysis
Many cancer cells have weakened interferon signaling and defective antiviral responses, which makes them more permissive to viral infection than normal cells. Oncolytic HSV takes advantage of this:
- It infects a tumor cell and hijacks its replication machinery.
- Viral replication stresses the cell and activates death pathways.
- The cell eventually bursts (lyses), releasing thousands of new virions that infect nearby tumor cells.
This self‑propagating cycle is the core of how herpes simplex virus as an anticancer agent directly debulks tumors.
2. Innate immune activation
Viral RNA and DNA act as danger signals that trigger pattern recognition receptors in tumor and immune cells, such as Toll‑like receptors and cytosolic DNA sensors.
- These sensors induce type I interferons and pro‑inflammatory cytokines.
- Innate immune cells—natural killer (NK) cells, macrophages, dendritic cells—are recruited into the tumor.
- They attack infected tumor cells and begin processing tumor antigens for presentation to T cells.
This innate response both limits uncontrolled viral spread and helps convert an immunologically “cold” tumor into a “hot” one.
3. Adaptive antitumor immunity
Perhaps the most exciting part of using herpes simplex virus as an anticancer agent is systemic immunity:
- Dendritic cells that have eaten debris from lysed tumor cells present tumor peptides to T cells in lymph nodes.
- Tumor‑specific CD8⁺ and CD4⁺ T cells expand and circulate.
- These T cells can then attack both injected tumors and distant, uninjected metastases.
Clinical trials with the HSV‑1 product T‑VEC have documented regression of non‑injected lesions, strong evidence that herpes simplex virus as an anticancer agent can act like a personalized cancer vaccine.
How scientists engineer herpes simplex virus as an anticancer agent
Wild‑type HSV‑1 can be dangerous in its natural form, so it must be heavily engineered before it can be used in patients.
Deleting virulence and immune‑evasion genes
Common modifications in oncolytic HSV vectors include:
- ICP34.5 deletion
ICP34.5 is a neurovirulence factor that helps HSV resist host antiviral shutdown of protein synthesis. Removing it makes the virus much safer in normal neural tissue yet still capable of replicating in many tumor cells with defective stress responses. - ICP47 deletion
ICP47 blocks antigen presentation via the TAP transporter, helping wild‑type HSV escape CD8⁺ T‑cell recognition. Deleting ICP47 increases MHC class I presentation of viral and tumor antigens, which enhances immune recognition when herpes simplex virus as an anticancer agent infects tumors.
These deletions are central to T‑VEC and many next‑generation oncolytic HSV designs.
Inserting immune‑boosting payloads
Modern vectors also insert therapeutic genes into the HSV genome:
- GM‑CSF (granulocyte‑macrophage colony‑stimulating factor)
T‑VEC expresses human GM‑CSF, which recruits and matures dendritic cells inside the tumor, improving antigen presentation and T‑cell priming. - Cytokines and checkpoint modulators
Experimental HSV‑1 vectors carry interleukins (such as IL‑12), chemokines, or even fragments of immune checkpoint inhibitors to further remodel the tumor microenvironment.
These additions turn herpes simplex virus as an anticancer agent into a dual‑action therapy: a direct tumor killer and a local factory for immune‑stimulating molecules.
Advanced retargeting strategies
To make herpes simplex virus as an anticancer agent even more selective, researchers are exploring:
- Envelope protein changes so the virus prefers tumor‑specific receptors.
- microRNA‑responsive cassettes that silence viral genes in normal tissues but not in tumors.
- Multiple immune payloads to counteract complex tumor immunosuppression.
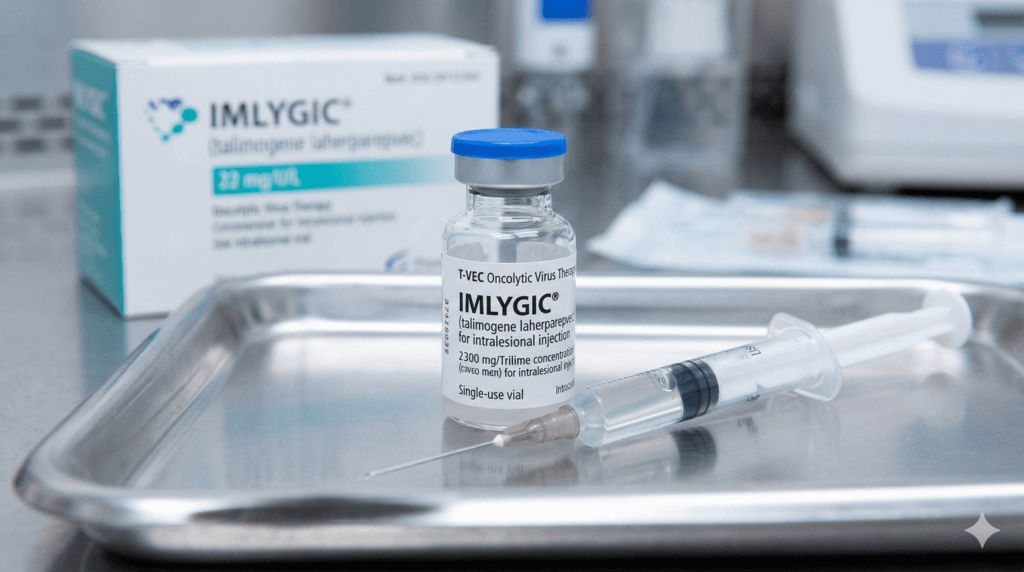
T‑VEC: the first approved herpes simplex virus as an anticancer agent
The strongest real‑world proof of concept for herpes simplex virus as an anticancer agent is T‑VEC (talimogene laherparepvec, Imlygic).
What is T‑VEC?
T‑VEC is a genetically modified HSV‑1 with:
- ICP34.5 deleted (to reduce neurovirulence and increase tumor selectivity)
- ICP47 deleted (to enhance antigen presentation)
- Human GM‑CSF inserted (to recruit and activate dendritic cells)
It is injected directly into melanoma lesions in the skin or lymph nodes and is currently approved for unresectable stage IIIB–IV melanoma with limited visceral disease.
Key clinical trial statistics (real‑world data)
The pivotal OPTiM phase III trial compared T‑VEC with GM‑CSF in patients with advanced melanoma.
Important outcomes:
- Durable response rate (DRR):
16.3% of patients treated with T‑VEC had responses lasting at least 6 months, compared with 2.1% in the GM‑CSF group—a significant improvement. - Overall responses and complete remissions:
T‑VEC produced higher overall response rates and a meaningful proportion of complete responses. A Melanoma Research Foundation summary reported that in a certain trial subgroup, 64% of patients responded and nearly half of those responses were complete, with many lasting over one year. - Safety profile:
Most side effects were flu‑like symptoms and injection‑site reactions; serious adverse events were relatively uncommon.
These results convinced the FDA to approve T‑VEC in 2015, making herpes simplex virus as an anticancer agent part of standard melanoma care for selected patients.
Cancers being targeted with herpes simplex virus as an anticancer agent
Although T‑VEC is only approved for melanoma, multiple clinical and preclinical programs are exploring herpes simplex virus as an anticancer agent across many malignancies.
Brain tumors and glioblastoma
Glioblastoma is an aggressive brain cancer with very poor prognosis despite surgery, radiation, and chemotherapy. HSV‑1 naturally infects neural tissues, so oncolytic HSV is a logical tool here.
- Early‑phase trials of HSV‑based vectors injected into recurrent glioblastoma have shown acceptable safety and occasional durable responses.
- Strategies include injection into the resection cavity and convection‑enhanced delivery to spread virus through infiltrative tumor regions.
While no HSV‑based drug is approved for glioblastoma yet, these studies demonstrate that herpes simplex virus as an anticancer agent can be safely delivered to the brain under controlled conditions.
Head and neck, gastrointestinal, and gynecologic cancers
Oncolytic HSV is being studied in:
- Head and neck squamous cell carcinoma
- Pancreatic cancer
- Colorectal cancer
- Ovarian and other gynecologic cancers
- Liver metastases and hepatocellular carcinoma
Preclinical work often shows synergy when herpes simplex virus as an anticancer agent is combined with chemotherapy or radiotherapy, improving tumor control compared with either modality alone.
Hematologic malignancies (early‑stage research)
There is early interest in using HSV‑1 against certain leukemias and lymphomas, often with ex vivo infection approaches or by targeting supportive stromal cells in the bone marrow niche. These ideas are still experimental but highlight how broadly researchers are thinking about herpes simplex virus as an anticancer agent.
Benefits, risks, and who can receive oncolytic HSV therapy
Like any potent cancer therapy, herpes simplex virus as an anticancer agent offers unique benefits and carries specific risks.
Major benefits
- Dual mechanism: direct tumor lysis plus systemic immune activation.
- Local delivery: intratumoral injection allows high local concentrations with limited systemic exposure (especially for T‑VEC).
- Potential for long‑term control: in some melanoma patients, T‑VEC has produced durable complete responses lasting years.
- Synergy with other treatments: preclinical and early clinical data show promising results when combined with chemotherapy, radiotherapy, and immune checkpoint inhibitors.
Common side effects
Most patients experience flu‑like symptoms and local reactions:
- Fever, chills, fatigue, headache
- Injection‑site pain, redness, or mild ulceration
- Mild nausea or malaise
These effects usually appear within the first few days after injection and resolve on their own.
Serious or theoretical risks
- Herpetic lesions or infections near the injection site or (rarely) in contacts if precautions fail.
- Higher risk in severely immunocompromised patients, who may not control viral replication as well and are often excluded from trials.
- Neurologic complications, although these have been rare thanks to ICP34.5 deletion and strict clinical protocols.
Overall, trials suggest that herpes simplex virus as an anticancer agent can be used safely when patients are properly selected and monitored.
Important: This article is for education only. Treatment decisions must always be made with a qualified oncologist.
Who is a candidate for T‑VEC right now?
For now, broad clinical use of herpes simplex virus as an anticancer agent is mainly limited to T‑VEC in melanoma.
Typical eligibility profile:
- Adults with unresectable cutaneous, subcutaneous, or nodal melanoma lesions after surgery.
- Limited visceral disease; T‑VEC is best for accessible lesions that can be injected.
- Adequate immune function and overall performance status.
Oncology teams often discuss whether to use T‑VEC alone or combined with systemic therapies like checkpoint inhibitors, depending on stage and risk factors.
For other cancers, herpes simplex virus as an anticancer agent is usually available only within clinical trials.
Future of herpes simplex virus as an anticancer agent
Researchers are already building next‑generation HSV‑1 vectors that go beyond T‑VEC.
Smarter targeting and deeper tumor penetration
New designs aim to:
- Retarget the virus to receptors enriched on tumor cells.
- Use tumor‑specific promoters and microRNA targeting to protect normal tissue.
- Improve spread through dense or poorly vascularized tumors.
Multi‑armored immunotherapy vectors
Future oncolytic HSV constructs may carry:
- Multiple cytokines (e.g., GM‑CSF plus IL‑12).
- Chemokines that attract specific immune subsets.
- Local expression of checkpoint inhibitors (e.g., anti‑PD‑1) inside the tumor to focus activity and reduce systemic toxicity.
This approach could make herpes simplex virus as an anticancer agent a powerful, localized immunotherapy platform.
Personalized combinations and biomarkers
The field is moving toward personalized viro‑immunotherapy:
- Tumor genomics and immune profiling may predict which patients benefit most from specific HSV constructs.
- Adaptive trials test combinations of HSV with PD‑1/PD‑L1 inhibitors, CTLA‑4 blockers, targeted drugs, and standard chemotherapy.
If successful, herpes simplex virus as an anticancer agent could become part of multi‑modal treatment plans tailored to each patient’s tumor and immune landscape.
Practical tips for patients interested in this therapy
If you or someone you know is considering herpes simplex virus as an anticancer agent, here are some actionable steps:
- Talk to your oncologist first
Ask whether T‑VEC or any oncolytic HSV trial might be appropriate for your specific cancer type and stage. - Check major trial registries
Search official registries (e.g., national clinical trial databases) for “oncolytic herpes simplex virus” or “HSV‑1 oncolytic virotherapy” plus your cancer type. - Look for comprehensive cancer centers
Large academic and research hospitals are more likely to run trials involving herpes simplex virus as an anticancer agent. - Ask about combinations
For melanoma, ask whether combining T‑VEC with an immune checkpoint inhibitor could be an option under current guidelines or trials. - Discuss logistics and safety
Inquire about how often injections are given, infection‑control precautions, and what symptoms to watch for.
Conclusion and future outlook
Herpes simplex virus as an anticancer agent has moved from theoretical concept to clinical reality, with the approval of T‑VEC marking a milestone in cancer immunotherapy and oncolytic virotherapy. Robust preclinical and clinical data show that HSV‑based vectors can selectively infect tumors, directly lyse cancer cells, and ignite systemic antitumor immune responses across multiple cancer types.
However, current HSV‑based therapies benefit only a subset of patients, and important scientific, logistical, and economic challenges remain. Future progress will likely come from rational vector engineering, biomarker‑guided patient selection, and intelligent combinations with other cancer treatments, especially immune checkpoint inhibitors and targeted agents.
If these hurdles can be overcome, herpes simplex virus as an anticancer agent may become a versatile platform integrated into multi‑modal cancer care, offering more patients durable control of their disease with an acceptable safety profile.
1. Is herpes simplex virus as an anticancer agent safe?
Current data suggest that herpes simplex virus as an anticancer agent, particularly T‑VEC, has a manageable safety profile in properly selected patients. Most side effects are flu‑like symptoms and mild injection‑site reactions, while serious complications are relatively uncommon under clinical supervision.
2. Can herpes simplex virus as an anticancer agent give me herpes?
T‑VEC is based on HSV‑1 but is genetically weakened and modified. Herpetic lesions at or near injection sites can occur, but strict handling precautions and dressings reduce the risk of spreading the virus to others. Clinicians can also use antiviral drugs if needed.
3. Which cancers can be treated with herpes simplex virus as an anticancer agent today?
Right now, T‑VEC is only approved for certain patients with unresectable melanoma involving skin and lymph nodes. Other uses of herpes simplex virus as an anticancer agent—in brain tumors, head and neck cancer, pancreatic cancer, and more—are still being studied in clinical trials.
4. How is herpes simplex virus as an anticancer agent given to patients?
For T‑VEC, an oncologist injects the viral solution directly into tumor lesions every 2–3 weeks in a clinic setting. Other experimental HSV‑based therapies may involve injection into the brain, body cavities, or occasionally systemic administration, depending on the trial.
5. Can herpes simplex virus as an anticancer agent be combined with immunotherapy drugs?
Yes, combining herpes simplex virus as an anticancer agent with immune checkpoint inhibitors (like anti‑PD‑1 antibodies) is one of the hottest research areas. Early data in melanoma suggest that adding T‑VEC to checkpoint blockade may increase response rates, but larger trials are needed to confirm long‑term benefits
6. Will pre‑existing HSV‑1 infection stop this treatment from working?
Many adults already carry HSV‑1 antibodies, which might neutralize some viruses in the bloodstream and could limit systemic delivery of herpes simplex virus as an anticancer agent. However, because T‑VEC and similar therapies are usually given directly into tumors, pre‑existing immunity has not been a major safety issue and may not fully block efficacy.
7. How long does it take to see results from herpes simplex virus as an anticancer agent?
Responses to T‑VEC often appear after several injection cycles, sometimes taking months to fully develop as both viral oncolysis and immune responses build up. Some patients show gradual shrinkage of injected and distant lesions over time rather than rapid, dramatic responses.
8. Is herpes simplex virus as an anticancer agent a cure for cancer?
No current therapy, including herpes simplex virus as an anticancer agent, is a guaranteed cure. T‑VEC and other oncolytic HSV treatments can produce long‑lasting remissions in a subset of patients, particularly in melanoma, but many patients still need additional systemic therapies. Researchers are working on combinations and next‑generation vectors to increase cure rates.




1 thought on “7 Powerful Ways Herpes Simplex Virus as an Anticancer Agent Fights Cancer”